Stem Cell Therapy for Specific Conditions
Are More Stem Cells Better?: New Study Using High-Cumulative-Dose Stem Cell Therapy for Idiopathic Pulmonary Fibrosis
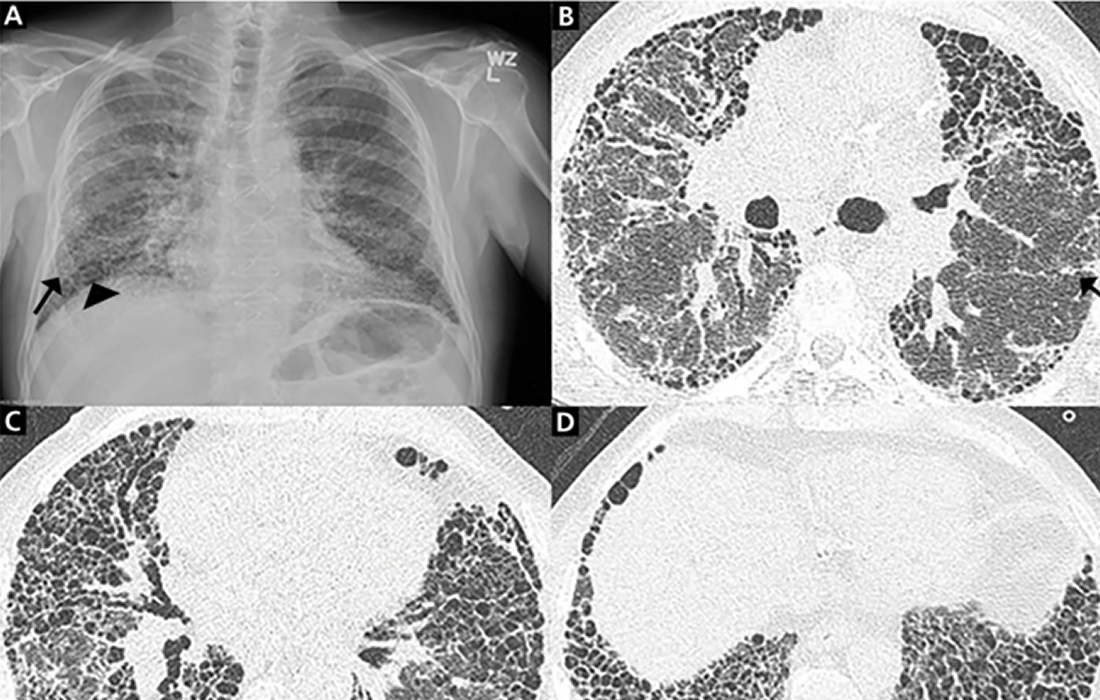
What is Pulmonary Fibrosis?
Pulmonary fibrosis (PF) is a chronic, life-threatening disease with a gradual worsening of pulmonary function and shortness of breath, and the median survival time of patients with idiopathic pulmonary fibrosis (IPF) was estimated to be 2.5–3.5 years.
Pulmonary fibrosis is a lung disease that occurs when lung tissue becomes damaged and scarred. This thickened, stiff tissue makes it more difficult for your lungs to work properly. As pulmonary fibrosis worsens, you become progressively more short of breath. The scarring associated with pulmonary fibrosis can be caused by a multitude of factors. But in most cases, the cause is unknown. When a cause can’t be found, the condition is termed idiopathic pulmonary fibrosis.
The prevalence of PF appears to be increasing, potentially explained by population aging.
What Causes PF?
Pulmonary fibrosis scars and thickens the tissue around and between the alveoli in the lungs, which makes the oxygen pass into the bloodstream difficult. It can be caused by many different factors, including long-term exposure to certain toxins, some medical conditions, radiation therapy, and some medications.
New Study Results
After a large number of successful preclinical studies, treatment with mesenchymal stem cells (MSCs) is considered a potential new direction for lung fibrosis therapy. The potential effects of MSCs in pulmonary fibrosis are related to their ability to produce a large number of biologically active substances with anti-inflammatory, immunosuppressive, and angiogenic properties.
In a new study published in the journal Stem Cells Translational Medicine, researchers evaluated the safety and tolerability of repeated infusions of high doses of bone marrow-derived MScs up to the total cumulative dose of 2 billion cells in subjects with rapidly progressing idiopathic pulmonary fibrosis.
The evaluation was based on the number and severity of adverse effects related to the infusion during 52 weeks of follow-up, during which no significant adverse effects. The only reported adverse effect was a transitory fever during the first day after the infusion noted in 4 out of 10 patients.
The researchers use intravenous MSCs transplantation as a method of delivery. The study included 20 patients that had forced ventilation capacity (FVC) ≥40% and diffusing capacity of the lung for carbon monoxide (DLCO) ≥20% with a decline of both >10% over the previous 12 months. They were randomized into 2 groups: one group received two intravenous doses of allogeneic MSCs (2 × 108 cells) every 3 months, and the second group received a placebo.
The group receiving the MSCs therapy had a significant improvement for the 6-minute walk distance in 13 weeks, for DLCO in 26 weeks, and for FVC in 39 weeks compared with the placebo group.
The therapy group had an increased 7.8% in FVC from baseline, whereas it declined by 5.9% in the placebo group.
Conclusions
The results of the study, according to the researchers which is the first-in-human clinical trial revealed that a high cumulative dose of MSCs is safe and well-tolerated by patients with PF with rapid lung function decline.
The patients that received the treatment had an increased lung function, while the patients in the placebo group experienced a continued decline in lung function, which shows the safety, tolerability, and potential benefits of using higher doses of MSCs.
Sources:
Averyanov A, Koroleva I, Konoplyannikov M, et al. First-in-human high-cumulative-dose stem cell therapy in idiopathic pulmonary fibrosis with rapid lung function decline. Stem Cells Transl Med. 2020;9(1):6-16. doi:10.1002/sctm.19-0037
https://www.mayoclinic.org/diseases-conditions/pulmonary-fibrosis/symptoms-causes/syc-20353690
https://my.clevelandclinic.org/health/diseases/10959-pulmonary-fibrosis
Image from: