Regenerative Medicine News and General Information
Fecal Microbiota Transplantation for Multiple Sclerosis Treatment
Multiple Sclerosis (MS) is an autoimmune, inflammatory, demyelinating disease of the central nervous system influenced by genetic susceptibility and environmental factors.
The gut microbiota is one such environmental factor that has been implicated in the development and progression of the disease. Previous studies have demonstrated that MS patients have numerous taxonomic alterations in their gut microbiota composition, including relative increases in Pseudomonas, Blautia, Streptococcus, and Akkermansia spp., and a decrease in Prevotella, Bacteroides, Parabacteroides, and Clostridia spp. compared to healthy individuals.
The gut microbiota can regulate intestinal permeability. With the alteration in the microbiota, increased intestinal permeability may allow microbial and dietary antigens to pass through the intestinal epithelium unregulated, leading to autoimmune responses. According to studies 20-70% of MS patients have elevated intestinal permeability.
In a recently published study, researchers evaluated the use of fecal microbiota transplantation in MS patients, and if it could improve intestinal permeability. The results appear in the Multiple Sclerosis Journal Experimental, Translational and Clinical.
Correction of Intestinal Permeability
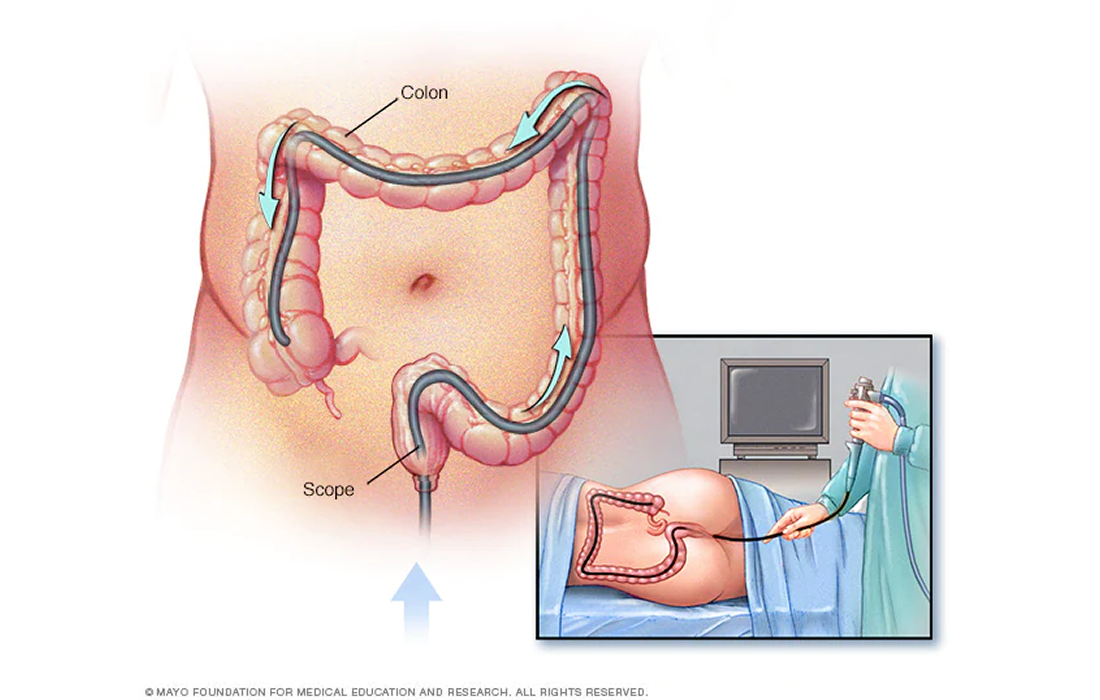
The study included a total of 9 patients with MS that were provided with monthly fecal microbiota transplantation for up to 6 months.
Stool samples of 50–70 g were collected from donors and stored as a whole stools at −80 °C for up to three months. The stool samples were thawed in a 37 °C water bath for one hour prior to the preparation of the rectal enema.
No major side effects were reported during the study. The treatment was safe and well-tolerated. The team observed that intestinal permeability was normalized, which could potentially help in the treatment of multiple sclerosis by decreasing the autoimmune response caused by different microbial and dietary antigens.
Source:
Kait F. Al, et al. Fecal microbiota transplantation is safe and tolerable in patients with multiple sclerosis: A pilot randomized controlled trial. 2022. MSJ Experimental, Translational and Clinical. https://doi.org/10.1177/20552173221086662.
Image from:
Mayo Foundation for Medical Education and Research. Retrieved from:
https://www.mayoclinic.org/es-es/tests-procedures/colonoscopy/about/pac-20393569