Regenerative Medicine News and General Information
New Treatment for Multiple Myeloma
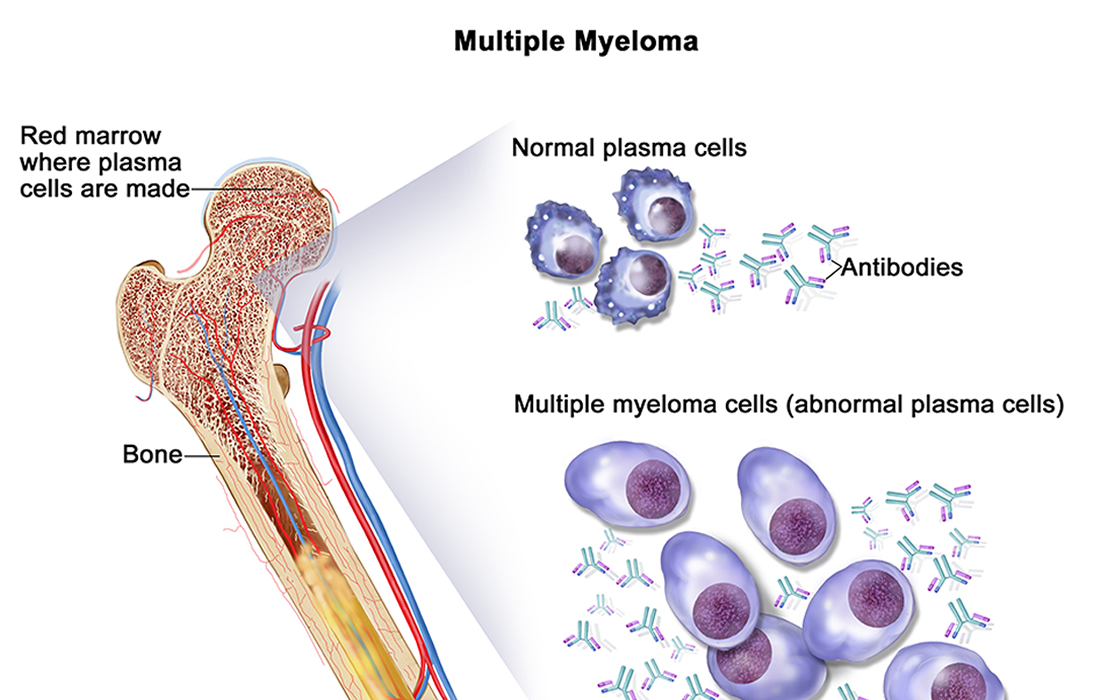
Multiple myeloma is a cancer that forms in a type of white blood cell called a plasma cell. Healthy plasma cells help you fight infections by making antibodies that recognize and attack germs.
In multiple myeloma, cancerous plasma cells accumulate in the bone marrow and crowd out healthy blood cells. Rather than produce helpful antibodies, the cancer cells produce abnormal proteins that can cause complications.
Current therapeutic options for multiple myeloma include immunomodulators, proteasome inhibitors, anti-CD38 monoclonal antibodies, chemotherapy, and corticosteroids.
Recently, the Food and Drug Administration (FDA) approved ciltacabtagene autoleucel (cita-cel) therapy to treat multiple myeloma in people who relapsed or did not respond to 3 or more therapies.
The new treatment is the second chimeric antigen receptor (CAR T cell) therapy approved, which consists in removing T cells from the blood of the person with multiple myeloma through a process called leukapheresis. During this process the blood passes through a machine that removes the T cells and infuses the remaining blood back into the patient.
The T cells are frozen and altered in the laboratory so that they have receptors that bind to specific proteins on cancer cells.
Study Results
The study enrolled participants 18 and older with multiple myeloma who failed 3 or more treatments, including a total of 97 participants.
Participants receiving cilta-cel showed a greater depth of response over time, with a complete response and normal bone marrow markers increasing from 67% at about 12 months to 83% at 22 months.
The 2-year survival without worsening multiple myeloma was about 61%, and the overall survival rate was 74%.
Sources:
Loli Uildriks. (2022, Mar 1). FDA approves cilta-cel for multiple myeloma. Medical News Today. Retrieved from:
https://www.medicalnewstoday.com/articles/fda-approves-cilta-cel-for-multiple-myeloma
https://www.mayoclinic.org/diseases-conditions/multiple-myeloma/symptoms-causes/syc-20353378
Image from:
https://www.cdc.gov/cancer/myeloma/index.htm